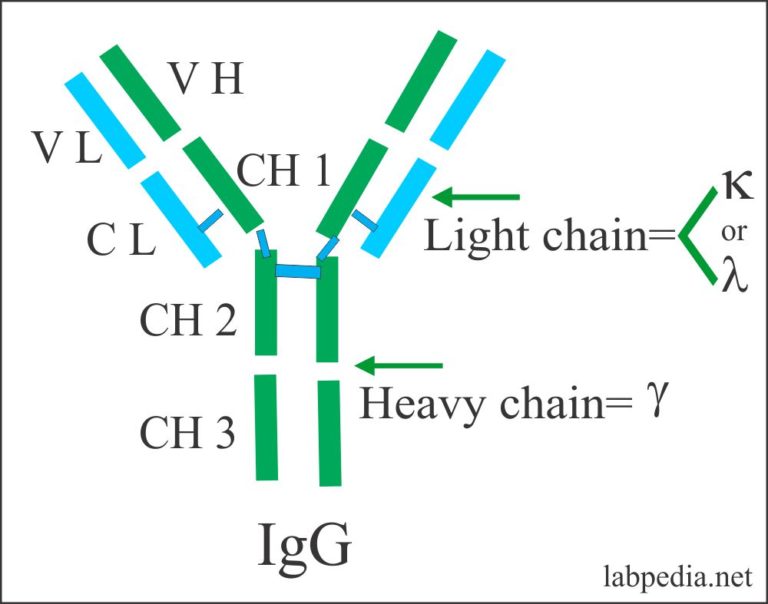
(2005) Divide and conquer: Division of labor by B-1 B cells. Journal of Experimental Medicine, 50, 533-550.Īlugupalli, K.R. Immuno-logical specificity of synthetic sugar-protein antigens. (1929) Chemo-immunological studies on conjugated carbohydrate proteins. (2003) Decline in invasive pneumococcal disease after the introduction of protein-polysaccharide conjugate vaccine. Whitney, C.G., Farley, M.M., Hadler, J., Harrison, L.H., Bennett, N.M., Lynfield, R., Reingold, A., Cieslak, P.R., Pilishvili, T., Jackson, D., Facklam, R. Morbidity and Mortality Weekly Report, 46, 1-24. (1997) Prevention of pneumococcal disease: Recommendations of the Advisory Committee on Immunization Practices (ACIP). These studies suggest that antigen recognition by polyreactive antibodies is determined by a conserved variable light chain CDR3 length and longer, more flexible variable heavy CDR3s when compared to pneumococcal polysaccha-ride-specific sequences while differences in specific avidities are modulated by antibody isotype. To further document the importance of the constant region in antibody avidity and fine specificity, analysis of antibody F(ab)’2 fragment binding to PPS14 and PPS23F resulted in similar K D values. The IgG1 antibodies uniformly had a stronger avidity to PPS14 and PPS23F compared to IgG2. To investigate the contribution of the constant region of these isotypes and their effect on antibody avidity to pneumococcal polysaccharide, the polyreactive variable regions were expressed as IgG1 or IgG2 and subjected to kinetic analysis. Variable chains are responsible for antigen recognition whereas antibody fine specificity is affected by isotype structure. While these antibodies were polyreactive and structurally alike, kinetic analysis revealed unique K D values. Most antibodies possessed a variable light chain with a CDR3 length made up of nine amino acids and relatively high number of flexible amino acids in combined VH/VL. Although isolated individually, these antibodies demonstrated similar characteristics. These antibodies were isolated from single pneumococcal polysaccharide specific B cells allowing for the analysis of human immunoglobulins with natively paired variable regions. We analyzed human polyreactive antibodies that bind multiple pneumococcal polysaccharides, including PPS14 and PPS23F. 
Polyreactive human pneumococcal polysaccharide IgG antibodies have not been extensively studied. Natural antibodies serve as the body’s first line of defense against pneumococcal challenge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
